 Moseley discovered that the number of protons in an element determines its atomic number.ġ919 Francis William Aston used a mass spectrograph to identify 212 isotopes.ġ922 Niels Bohr proposed an atomic structure theory that stated the outer orbit of an atom could hold more electrons than the inner orbit.ġ923 Louis de Broglie proposed that electrons have a wave/particle duality.ġ929 Cockcroft / Walton created the first nuclear reaction, producing alpha particlesġ930 Paul Dirac proposed the existence of anti-particles.ġ932 James Chadwick discovered neutrons, particles whose mass was close to that of a proton.ġ938 Lise Meitner, Hahn, Strassman discovered nuclear fission.ġ941-51 Glenn Seaborg discovered eight transuranium elements.ġ942 Enrico Fermi created the first man-made nuclear reactor.\): Dalton's symbols. Atomic Model (Atomic Theory) - An atom is the smallest unit of ordinary matter that forms a chemical element.Atoms are made of fundamental particles called protons, neutrons and electrons. Thomson determined the charge to mass ratio of electrons.ġ898 Rutherford discovered alpha, beta, and gamma rays in radiation.ġ898 Marie Sklodowska Curie discovered radium and polonium and coined the term radioactivity after studying the decay process of uranium and thorium.ġ900 Max Planck proposed the idea of quantization to explain how a hot, glowing object emitted light.ġ900 Frederick Soddy came up with the term “isotope” to explain the unintentional breakdown of radioactive elements.ġ903 Hantaro Nagaoka proposed an atomic model called the Saturnian Model to describe the structure of an atom.ġ904 Richard Abegg found that inert gases have a “stable electron configuration.”ġ906 Hans Geiger invented a device that could detect alpha particles.ġ914 H.G.J. Chadwick was educated at the University of Manchester, where he worked under Ernest Rutherford and earned a master’s degree in 1913. 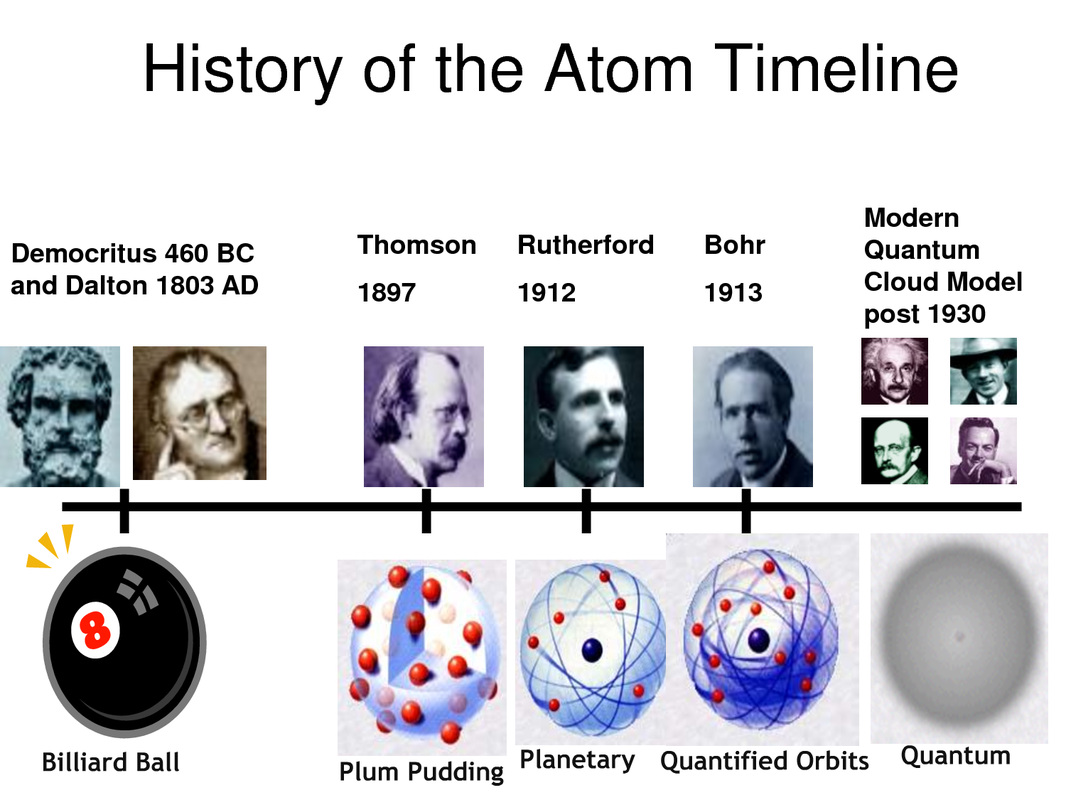
The current model, the Electron Cloud Model. Even today we’re not entirely sure but we have a far. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called electrons, circulate at some distance, much like planets revolving around the Sun. James Chadwick (born October 20, 1891, Manchester, Englanddied July 24, 1974, Cambridge, Cambridgeshire) English physicist who received the Nobel Prize for Physics in 1935 for the discovery of the neutron. Atomic theory is the collection of ideas that have morphed over time as more and more information about the structure of the atom has become clearer. Goldstein discovered canal rays, which have a positive charge equal to an electron.ġ896 Henri Becquerel discovered radiation by studying the effects of x-rays on photographic film.ġ897 J.J. Single-atom alloys have emerged as highly active and selective catalysts that do not follow the traditional models of heterogeneous catalysis. Atomic theory Matter Physics FuseSchoolWe didn’t always know exactly what the atom looked like. Many teachers relate the different historical understandings of atomic structure to different varieties of candy. Stoney theorized that electricity was comprised of negative particles he called electrons.ġ879 Sir William Crookes’ experiments with cathode-ray tubes led him to confirm the work of earlier scientists by definitively demonstrating that cathode-rays have a negative charge.ġ886 E. Though we know today that they can be further divided into protons, neutrons. 
Plucker built one of the first cathode-ray tubes.ġ869 Dmitri Mendeleev created the periodic table.ġ873 James Clerk Maxwell proposed the theory of electromagnetism and made the connection between light and electromagnetic waves.ġ874 G.J. He defined an atom as the smallest indivisible particle. /GettyImages-141483984-56a133b65f9b58b7d0bcfdb1.jpg) 
According to the model, the nucleus is a very. Democritus’ atomic theory posited that all matter is made up small indestructible units he called atoms.ġ704 Isaac Newton theorized a mechanical universe with small, solid masses in motion.ġ803 John Dalton proposed that elements consisted of atoms that were identical and had the same mass and that compounds were atoms from different elements combined together.ġ832 Michael Faraday developed the two laws of electrochemistry.ġ859 J. The Rutherford atomic model has 2 main parts: the nucleus, and the atoms remaining space, occupied by electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
